Are you a US healthcare professional?
INDICATION
RYTELO® (imetelstat) is indicated for the treatment of adult patients with low- to intermediate-1 risk myelodysplastic syndromes (MDS) with transfusion-dependent anemia requiring 4 or more red blood cell units over 8 weeks who have not responded to or have lost response to or are ineligible for erythropoiesis-stimulating agents (ESA). See more
RYTELO safety profile
from the IMerge trial1,2,*
Adverse reactions commonly reported in the IMerge phase 3 trial
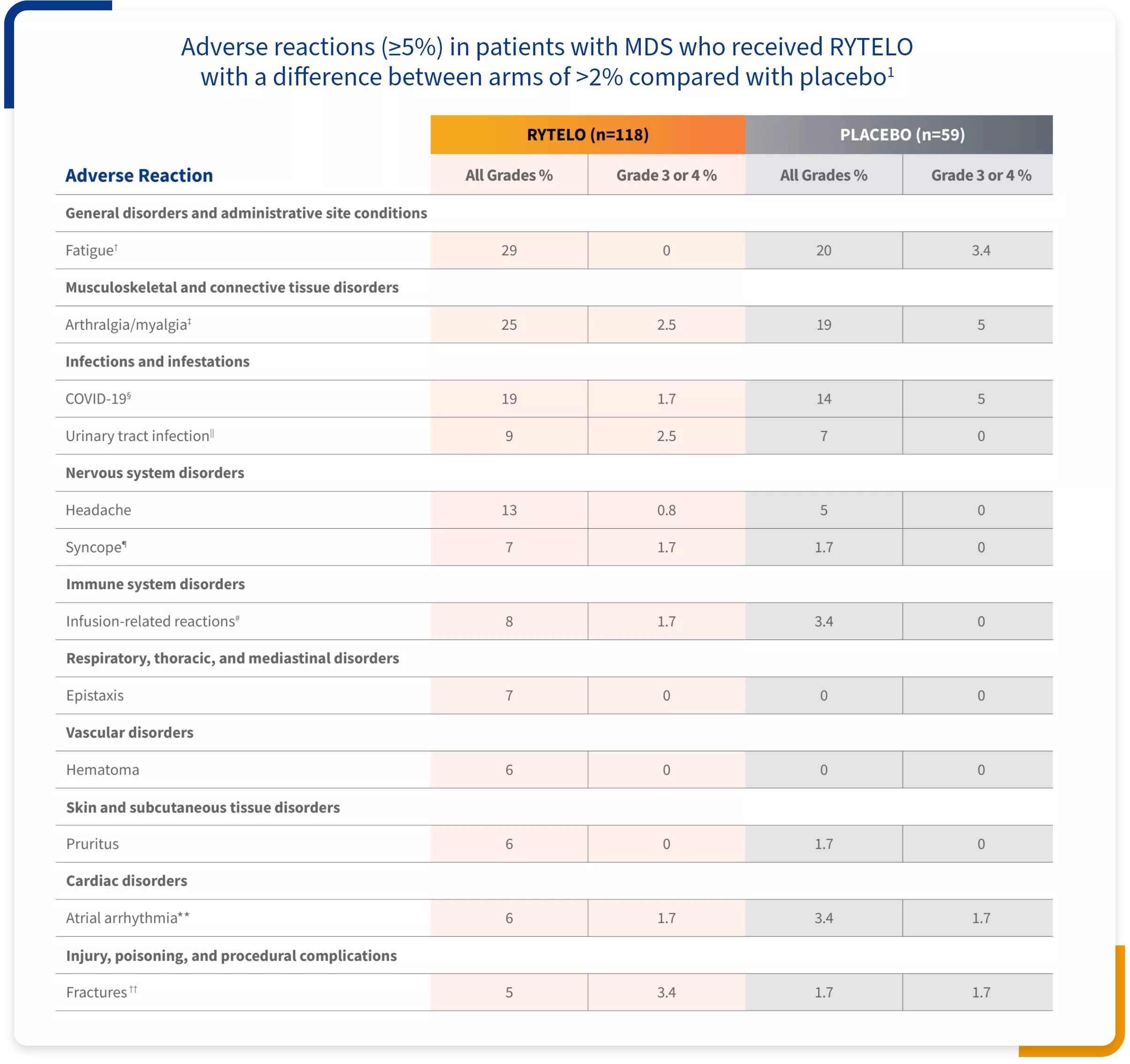
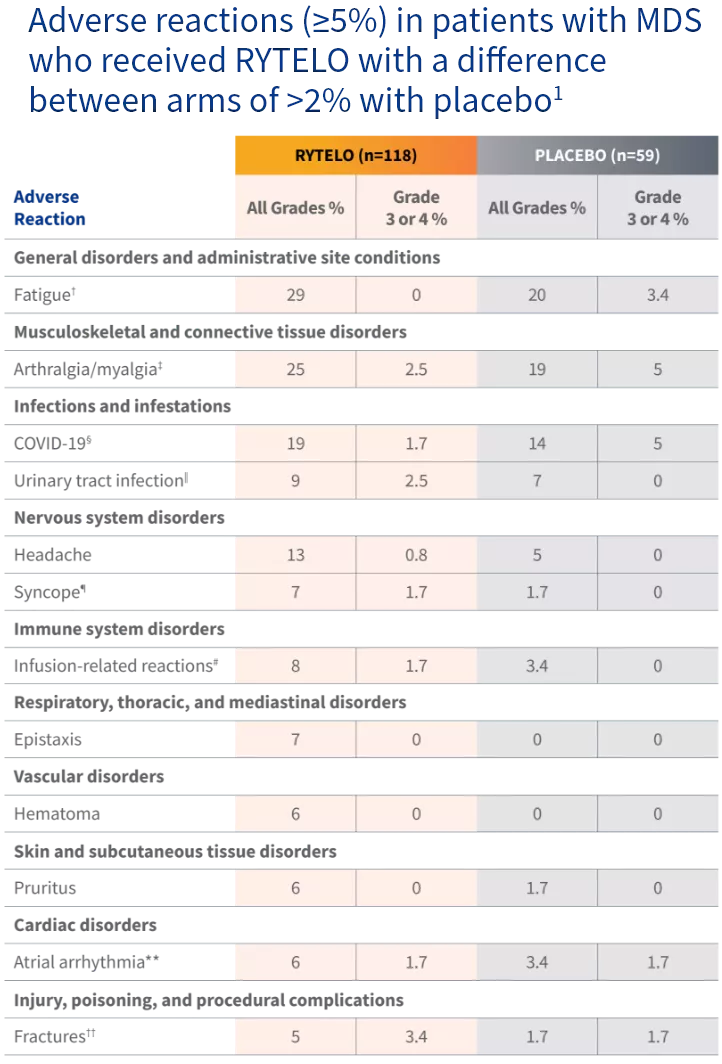
of patients receiving RYTELO compared to 3% (n=2) of patients on placebo1
Graded according to National Cancer Institute Common Terminology Criteria for Adverse Events Version 4.03.
*All presented safety data within: Data cutoff May 10, 2023.
†Fatigue: asthenia, fatigue, and malaise.
‡Arthralgia/myalgia: arthralgia, back pain, bone pain, musculoskeletal pain, myalgia, neck pain, non-cardiac chest pain, pain, pain in extremity, pain in jaw, and pelvic pain.
§COVID-19: asymptomatic COVID-19, COVID-19, COVID-19 pneumonia, and SARS-CoV-2 antibody test positive.
||Urinary tract infection: cystitis, Escherichia urinary tract infection, renal abscess, and urinary tract infection.
¶Syncope: fall, pre-syncope, and syncope.
#Infusion-related reactions: abdominal pain, arthralgia, asthenia, back pain, bone pain, diarrhea, erythema, headache, hypertensive crisis, malaise, non-cardiac chest pain, pruritus, and urticaria. Only events considered related to infusion-related reactions are included.
**Atrial arrhythmia: atrial fibrillation and atrial flutter.
††Fractures: femur fracture, hand fracture, hip fracture, humerus fracture, lumbar vertebral fracture, and thoracic vertebral fracture.
Select laboratory abnormalities observed with RYTELO
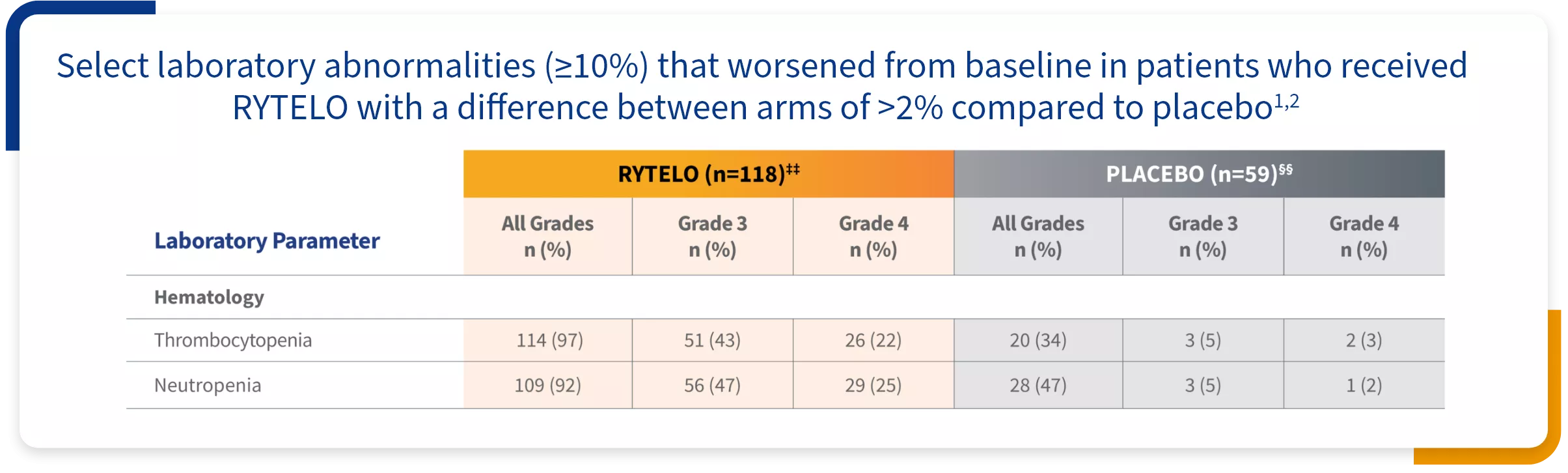
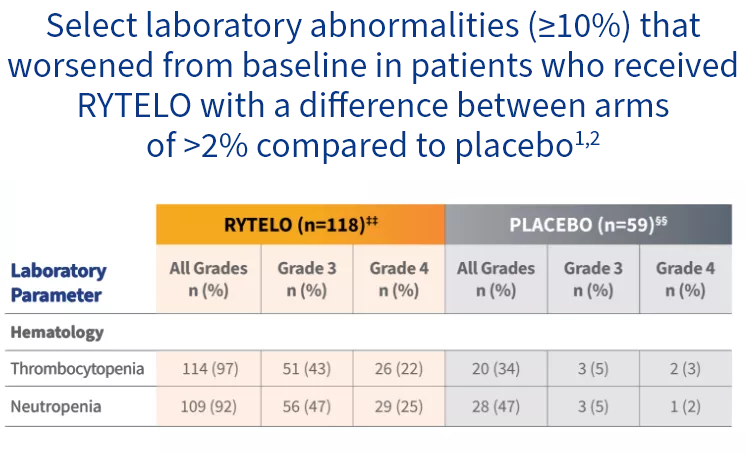
Graded according to National Cancer Institute Common Terminology Criteria for Adverse Events Version 4.03.
‡‡The denominator used to calculate the rate varied from 97 to 118 based on the number of patients with a baseline value and at least 1 posttreatment value.1
§§The denominator used to calculate the rate varied from 50 to 59 based on the number of patients with a baseline value and at least 1 posttreatment value.1
Grade ≥3 clinical consequences of cytopenias were generally comparable to placebo2
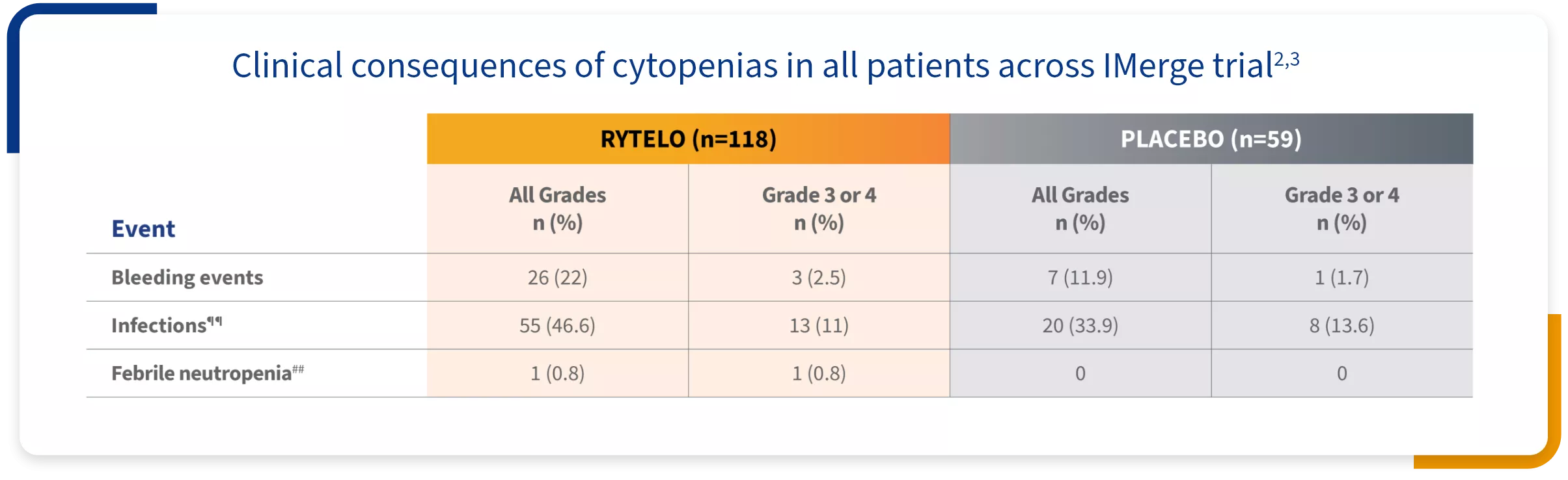
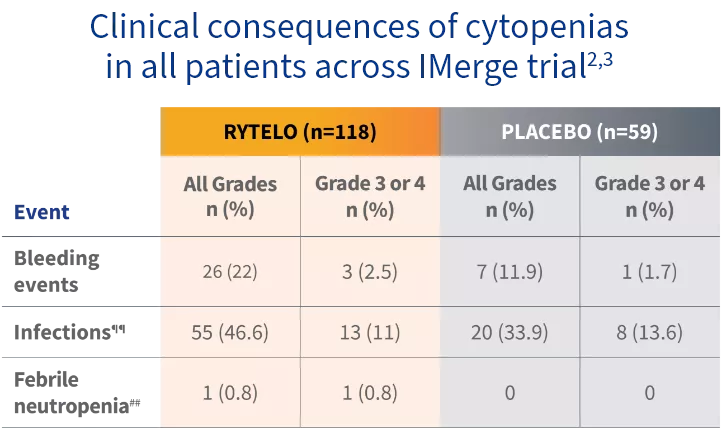
Supportive care:
- 17.8% (n=21) of RYTELO-treated patients received a median of 1 platelet transfusion vs 1.7% (n=1) on placebo2
- 34.7% (n=41) of RYTELO-treated patients received growth factor support vs 3.3% (n=2) on placebo2
¶¶Three patients in the RYTELO group had Grade 3 or 4 infections concurrent with Grade 3 or 4 neutropenia.
##Occurred at day 33, lasted 8 days; assessed by investigator as possibly related to RYTELO; patient subsequently achieved TI >40 weeks and remained on treatment until data cutoff.
treatment were associated with increased Hgb from pre-treatment4,5,†††,‡‡‡,■
■Limitations: This post hoc analysis was descriptive in nature, not prespecified, and did not control for type 1 error. Given the post hoc nature of this analysis and the small sample sizes of the subgroups, interpret with caution as results could represent chance findings. This analysis was limited by differing methodologies of the phase 2, phase 3, and QTc substudy components of the IMerge trial.
***Patients with ≥50% maximum platelet reduction vs those with <50%; patients with ≥75% maximum neutrophil reduction vs those with <75%.
†††Post hoc pooled analysis (N=226) of patients treated with RYTELO 7.1 mg/kg IV infusion every 4 weeks from the 3 parts of IMerge; Phase 2 (n=57), Phase 3 (n=118), and the QTc substudy (n=51). Patients were: IPSS low- or intermediate-1–risk MDS, relapsed or refractory to ESAs or sEPO >500 mU/mL (ESA-ineligible) and transfusion dependent (requiring ≥4 RBC U/8 weeks over 16-week prestudy period). Differing methodologies were: patients with del(5q) or prior lenalidomide or HMA were included in the phase 2 and QTc substudy. Patients in the QTc substudy could cross over from placebo to RYTELO at the investigator’s discretion.5
‡‡‡Hgb values within 14 days of RBC transfusion were excluded due to impact of transfusions.
Grade ≥3 cytopenias: Expected and generally short duration1,2
EARLY ONSET
The median time to onset of cytopenia events for patients taking RYTELO occurred during the first 2 cycles1
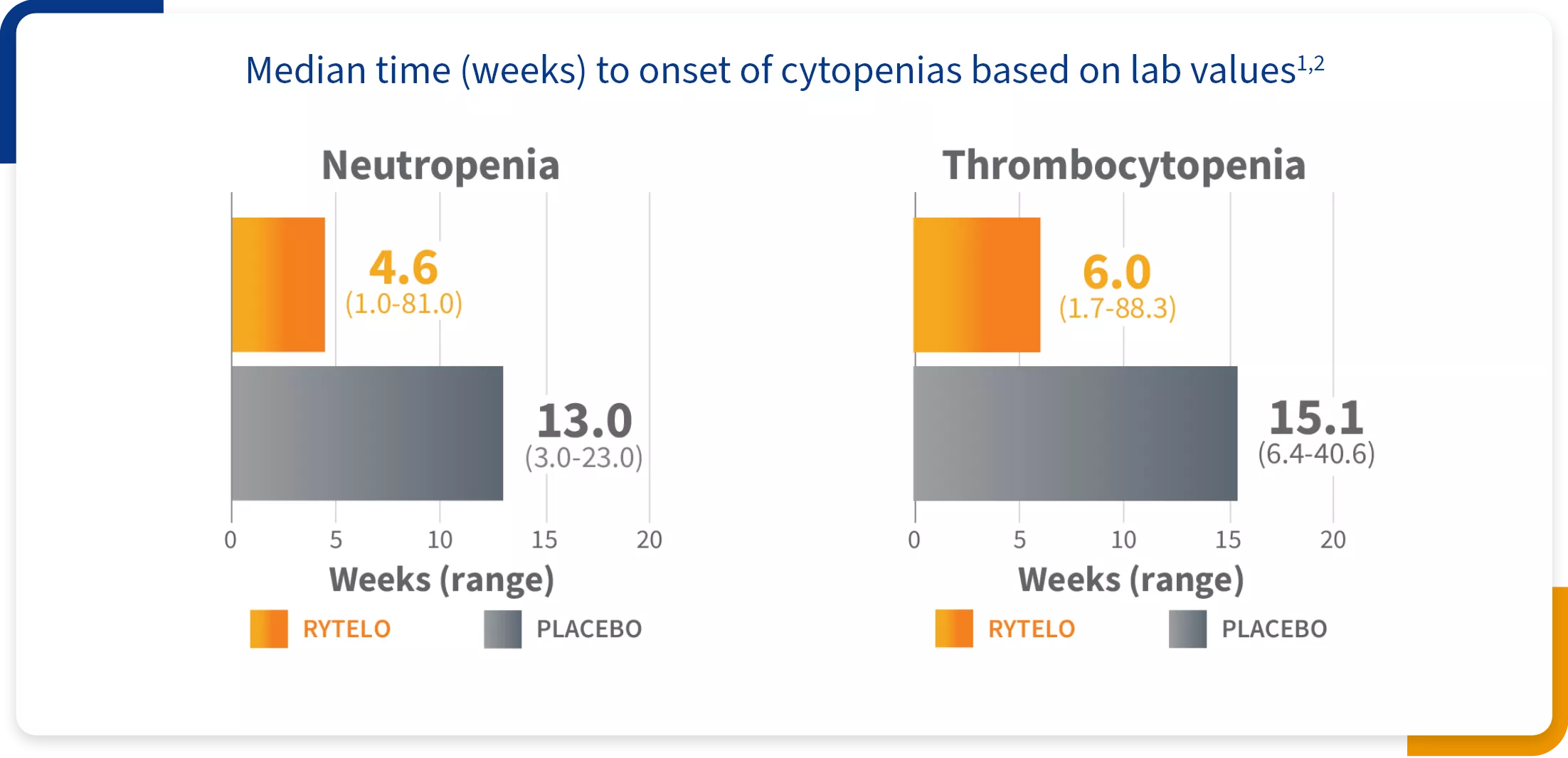
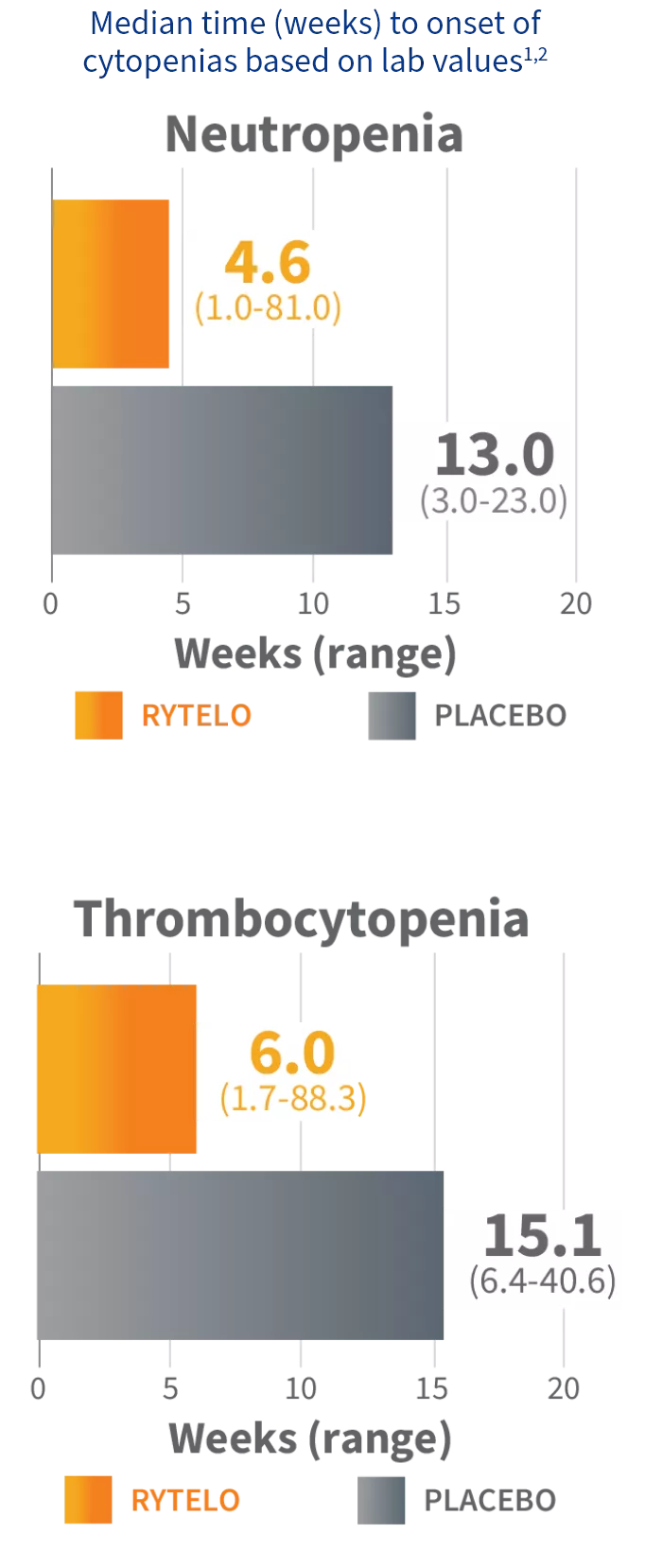
COMMONLY TRANSIENT
In >80% of events, platelets recovered to ≥50,000/mm3 and neutrophils recovered to ≥1000/mm3 within 4 weeks for patients on RYTELO2,§§§
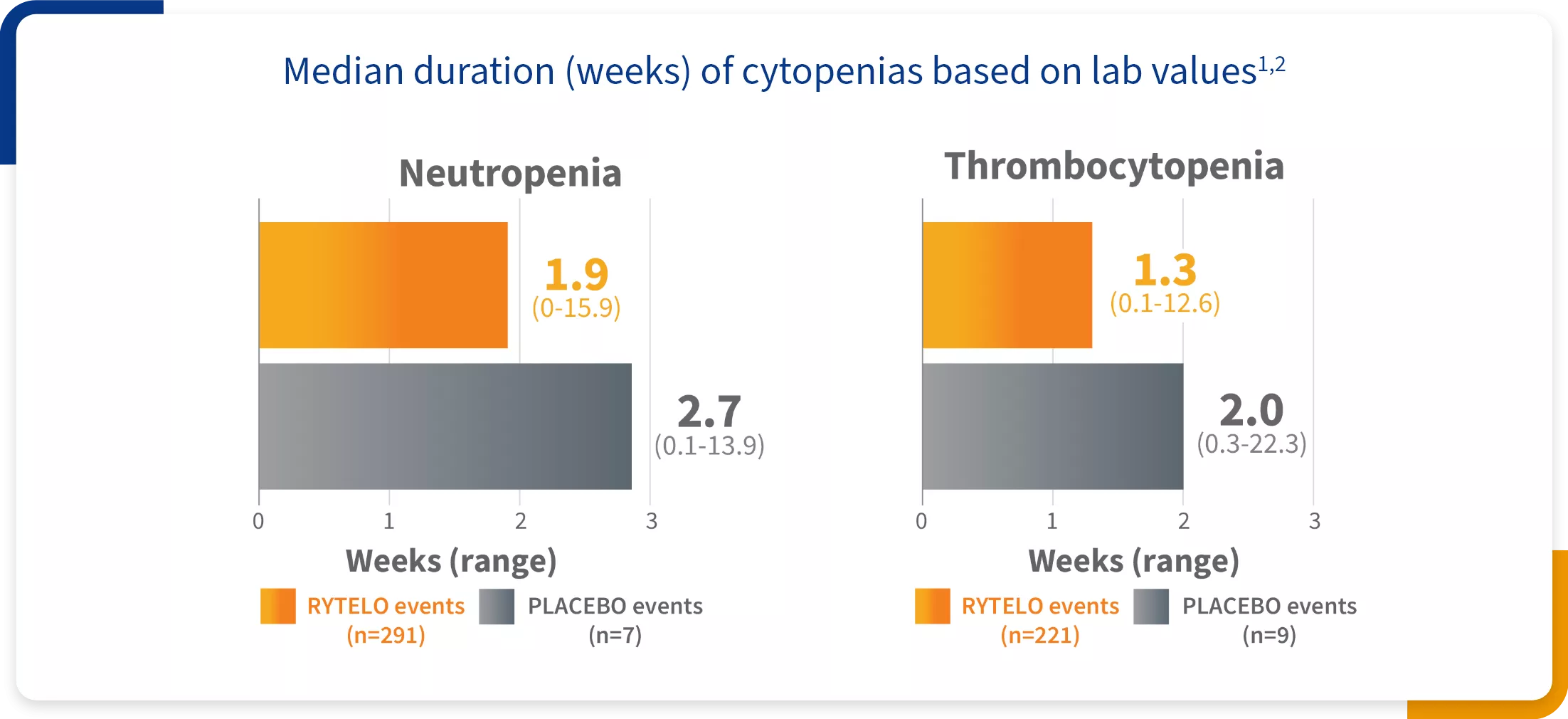
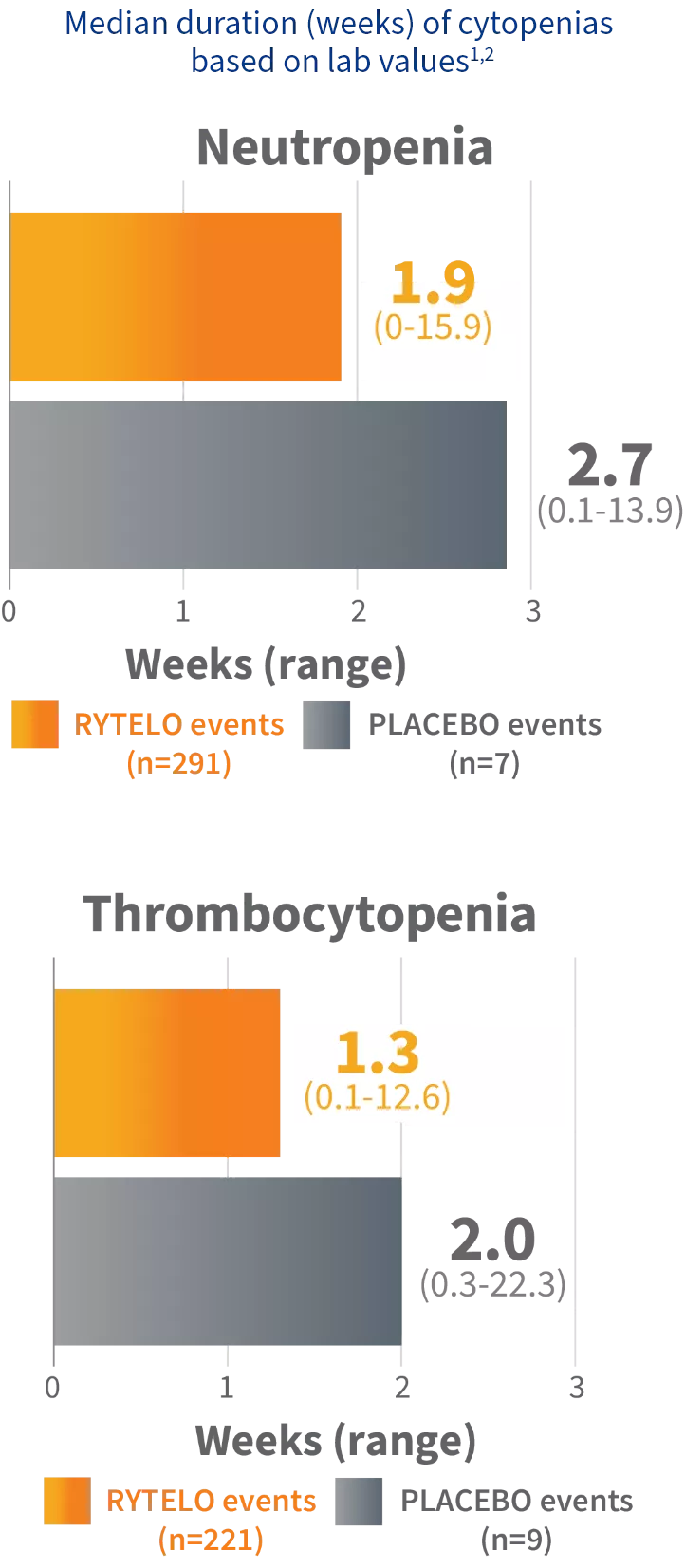
- Neutropenia events returned to Grade ≤2 within 4 weeks for 77% of patients taking RYTELO and 98% on placebo2
- Thrombocytopenia events returned to Grade ≤2 within 4 weeks for 90% of patients taking RYTELO and 95% on placebo2
§§§Recovery occurred due to dose delays or reductions. All data presented indicate CTCAE Grade ≥2.
INDICATION
RYTELO® (imetelstat) is indicated for the treatment of adult patients with low- to intermediate-1 risk myelodysplastic syndromes (MDS) with transfusion-dependent anemia requiring 4 or more red blood cell units over 8 weeks who have not responded to or have lost response to or are ineligible for erythropoiesis-stimulating agents (ESA).
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
Thrombocytopenia
RYTELO® can cause thrombocytopenia based on laboratory values. In the clinical trial, new or worsening Grade 3 or 4 decreased platelets occurred in 65% of patients with MDS treated with RYTELO.
Monitor patients with thrombocytopenia for bleeding. Monitor complete blood cell counts prior to initiation of RYTELO, weekly for the first two cycles, prior to each cycle thereafter, and as clinically indicated. Administer platelet transfusions as appropriate. Delay the next cycle and resume at the same or reduced dose, or discontinue as recommended.
Neutropenia
RYTELO can cause neutropenia based on laboratory values. In the clinical trial, new or worsening Grade 3 or 4 decreased neutrophils occurred in 72% of patients with MDS treated with RYTELO.
Monitor patients with Grade 3 or 4 neutropenia for infections, including sepsis. Monitor complete blood cell counts prior to initiation of RYTELO, weekly for the first two cycles, prior to each cycle thereafter, and as clinically indicated. Administer growth factors and anti-infective therapies for treatment or prophylaxis as appropriate. Delay the next cycle and resume at the same or reduced dose, or discontinue as recommended.
Infusion-Related Reactions
RYTELO can cause infusion-related reactions. In the clinical trial, infusion-related reactions occurred in 8% of patients with MDS treated with RYTELO; Grade 3 or 4 infusion-related reactions occurred in 1.7%, including hypertensive crisis (0.8%). The most common infusion-related reaction was headache (4.2%). Infusion-related reactions usually occur during or shortly after the end of the infusion.
Premedicate patients at least 30 minutes prior to infusion with diphenhydramine and hydrocortisone as recommended and monitor patients for at least one hour following the infusion as recommended. Manage symptoms of infusion-related reactions with supportive care and infusion interruptions, decrease infusion rate, or permanently discontinue as recommended.
Embryo-Fetal Toxicity
Based on animal findings, RYTELO can cause embryo-fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with RYTELO and for 1 week after the last dose.
ADVERSE REACTIONS
Serious adverse reactions occurred in 32% of patients who received RYTELO. Serious adverse reactions in >2% of patients included sepsis (4.2%), fracture (3.4%), cardiac failure (2.5%), and hemorrhage (2.5%). Fatal adverse reactions occurred in 0.8% of patients who received RYTELO, including sepsis (0.8%).
Most common adverse reactions (≥10% with a difference between arms of >5% compared to placebo), including laboratory abnormalities, were decreased platelets, decreased white blood cells, decreased neutrophils, increased AST, increased alkaline phosphatase, increased ALT, fatigue, prolonged partial thromboplastin time, arthralgia/myalgia, COVID-19 infections, and headache.
Please see full Prescribing Information, including Medication Guide.
You are encouraged to report adverse events related to Geron products by calling 1-855-437-6664 (1-855-GERON-MI) (US only). If you prefer, you may contact the US Food and Drug Administration (FDA) directly. Visit www.fda.gov/MedWatch or call 1-800-FDA-1088.


